"FDA Issues Recall for CVS and Walmart Eye Ointments Over Infection Risk"
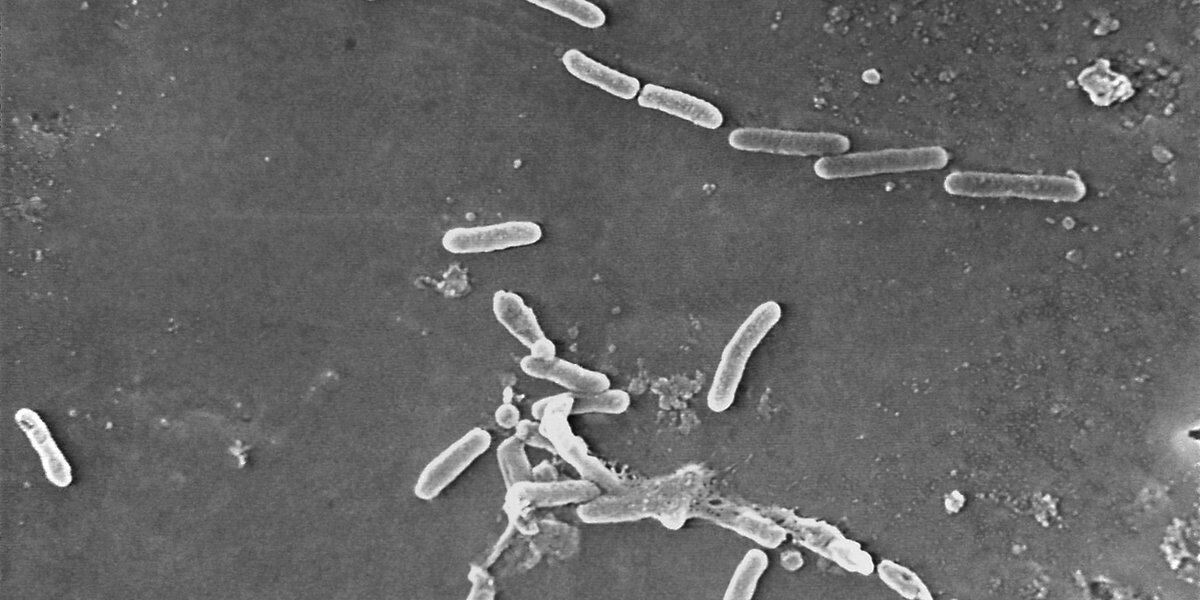
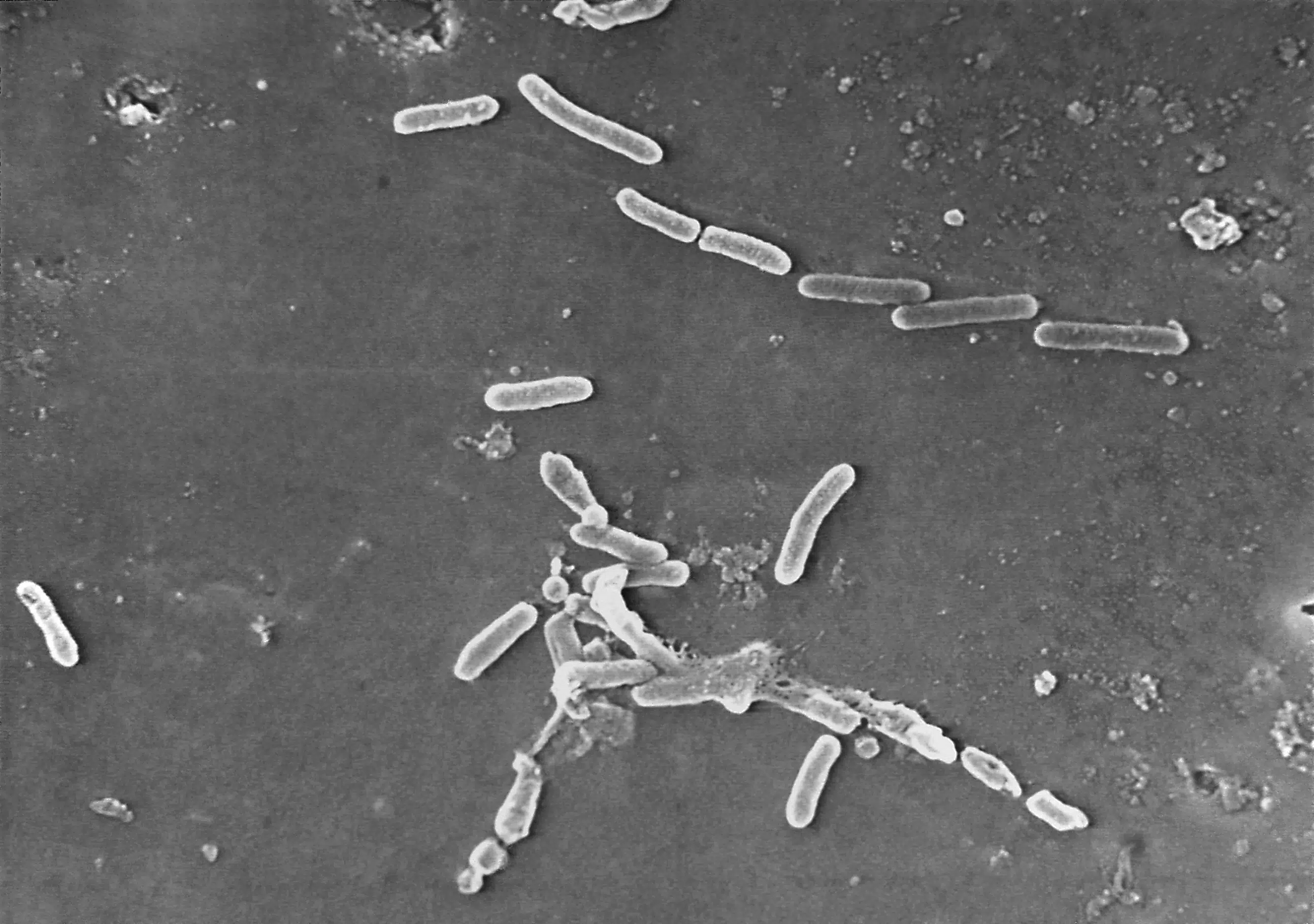
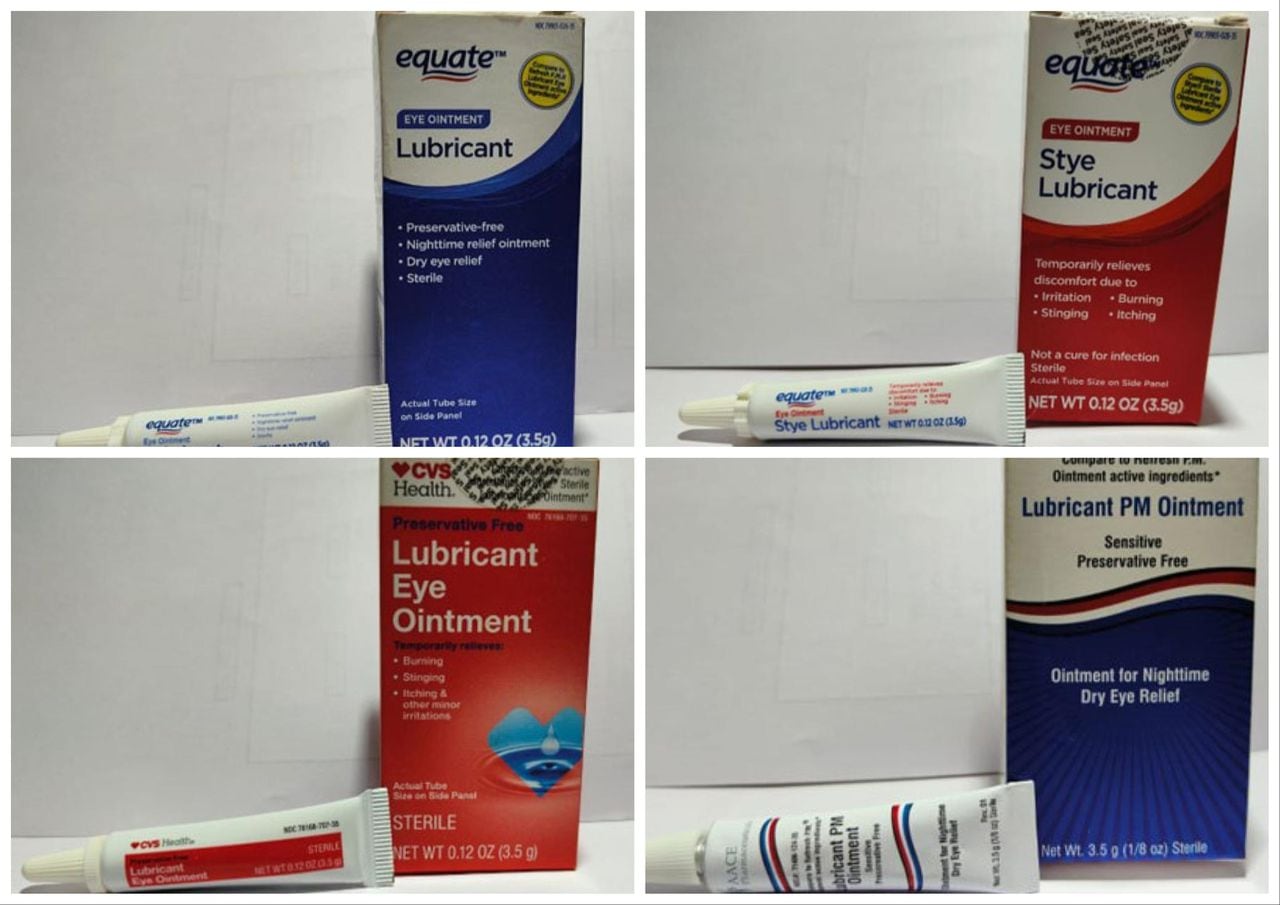
The FDA has issued a recall for four types of eyedrops sold at Walmart and CVS due to a lack of sterility assurance at the facility where they are made. The eyedrops, produced by Brassica Pharma Pvt. Ltd., are at risk of potential eye infections or related harm and should not be used. The recalled products should be returned to the retailer where they were purchased, and individuals can visit the FDA website for more information on the recall.