
Moderna's mRNA flu shot moves to FDA review after dispute resolution
Moderna says the FDA will review its experimental mRNA-based flu vaccine after resolving a public dispute, signaling movement toward potential regulatory consideration of the shot.
All articles tagged with #moderna

Moderna says the FDA will review its experimental mRNA-based flu vaccine after resolving a public dispute, signaling movement toward potential regulatory consideration of the shot.
The FDA reversed its initial refusal to accept Moderna’s seasonal flu vaccine filing after a White House meeting in which President Trump pressed the agency, with Moderna submitting an amended filing and the FDA agreeing to review it via a Type A meeting. The agency accepted the application for full approval for adults 50–64 and accelerated approval for 65+, with a post‑marketing study to address age-related concerns, and a target decision date of August 5, 2026.
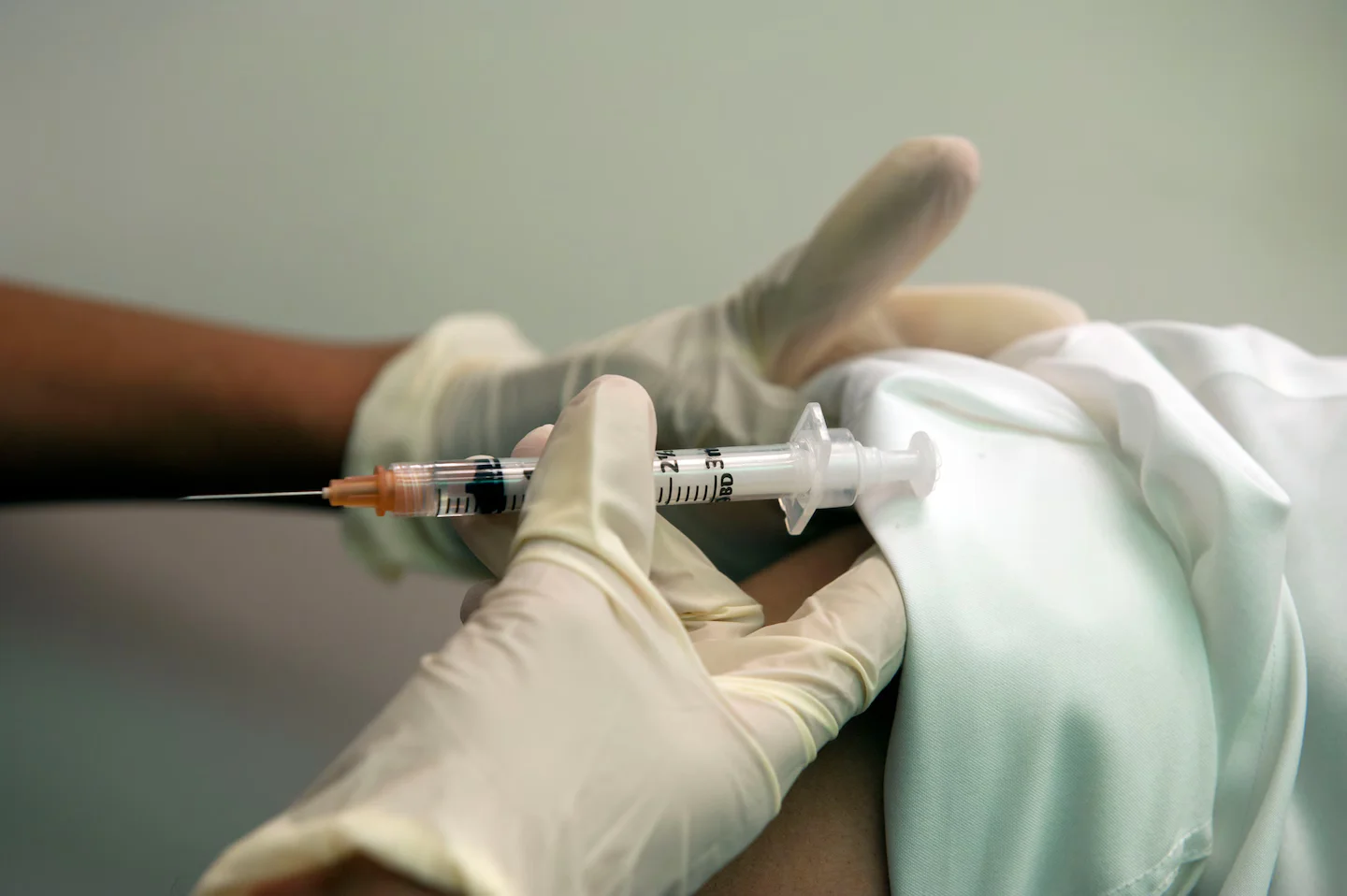
The FDA reversed its earlier decision and will review Moderna’s first mRNA-based influenza vaccine after Moderna agreed to conduct additional studies in older adults under a revised review process.

The FDA has agreed to review Moderna’s experimental mRNA flu vaccine after previously declining to file, with a decision due by August 5; Moderna seeks full approval for adults 50–64 and accelerated approval for 65+, contingent on a post‑marketing study, amid broader regulatory shifts and debate over vaccine oversight.

The FDA has reversed its earlier stance and agreed to review Moderna’s updated mRNA flu vaccine, with plans for full approval in adults 50–64 and accelerated approval for those 65 and older, plus a post-marketing study in older adults. A decision is expected by August 5, and Moderna shares rose on the news.

The FDA will review Moderna’s newly developed mRNA seasonal flu vaccine after a prior rejection over trial design, with no safety or efficacy concerns cited. Moderna says it has met with the FDA and proposed a revised regulatory pathway, seeking full approval for adults 50–64 and accelerated approval for 65+, potentially making the vaccine available for those 50+ in the 2026–2027 flu season if approved.

The FDA reversed its earlier decision and will review Moderna’s new mRNA flu vaccine for adults 50 and older, using a standard pathway for ages 50–64 and accelerated approval for those 65+, with a required post‑marketing study and a target review date of August 5.

Moderna is pulling back on late-stage vaccine studies, while other vaccine developers pause projects and cut positions amid a chilling policy environment under health secretary RFK Jr. The trend is illustrated by Moderna pausing trials in Massachusetts, a Texas factory project being canceled, and layoffs at a San Diego manufacturer, signaling a broader slowdown in vaccine development and manufacturing.

Moderna’s mRNA influenza vaccine was not even considered by the FDA after a “refuse to file” ruling, despite earlier signals that its trial design was acceptable. The decision is described as coming from Vinay Prasad, an FDA appointee aligned with RFK Jr., who overruled career staff, signaling a broader shift under Kennedy’s leadership away from standard scientific processes. The piece warns this regulatory pivot could deter vaccine investment, undermine pandemic preparedness, and slow the development of future mRNA vaccines.

The FDA declined to review Moderna’s experimental mRNA flu vaccine, citing an allegedly inadequate trial design for adults 65 and older that used a standard-dose control instead of a high-dose option, prompting critics to allege an anti-vaccine agenda and a chilling effect on vaccine development; Moderna and its supporters argue the design was approved and that higher-dose controls should have informed the analysis.

Moderna shares rose about 10% after Phase 2 data showed its cancer vaccine, when combined with Keytruda, reduced relapse or death risk in melanoma, signaling potential for its oncology pipeline as Covid-era vaccine sales fade and cash stockpiles dwindle. Evercore ISI called the asset a key value driver, while the stock remains rated a Hold by analysts with notable downside risk baked into targets.

Stock markets saw mixed movements with Nvidia rising 2% after licensing AI technology from Groq, Coupang surging 8.8% following an internal cybersecurity investigation, and Lam Research approaching record highs. Tesla declined 1.3% amid safety concerns, while Moderna dropped 4.6% as its pandemic vaccine stock continued to decline. Other notable movements included Nike, Micron, and Newmont, with the market volume remaining thin due to the holiday season.

Moderna's experimental cytomegalovirus (CMV) vaccine failed in a Phase 3 trial, marking a significant setback for the company, which had high sales expectations for the vaccine prior to the failure amid ongoing pressures from Wall Street and the government.
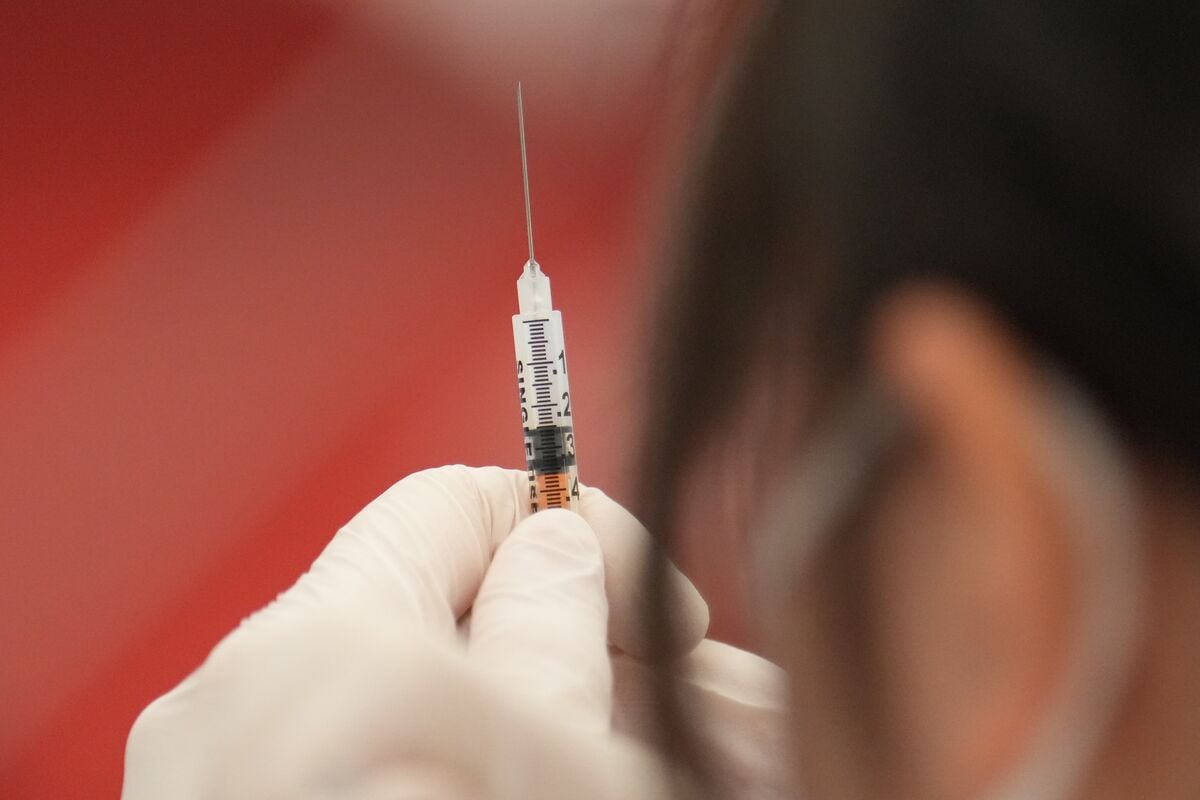
Shares of Moderna and other vaccine makers fell after reports suggested Trump health officials might link Covid vaccine data to the deaths of 25 children, raising concerns about vaccine safety despite scientific studies indicating rare serious side effects. The CDC and FDA continue to monitor vaccine safety, with ongoing investigations into adverse events, while the vaccines remain widely supported for their benefits.
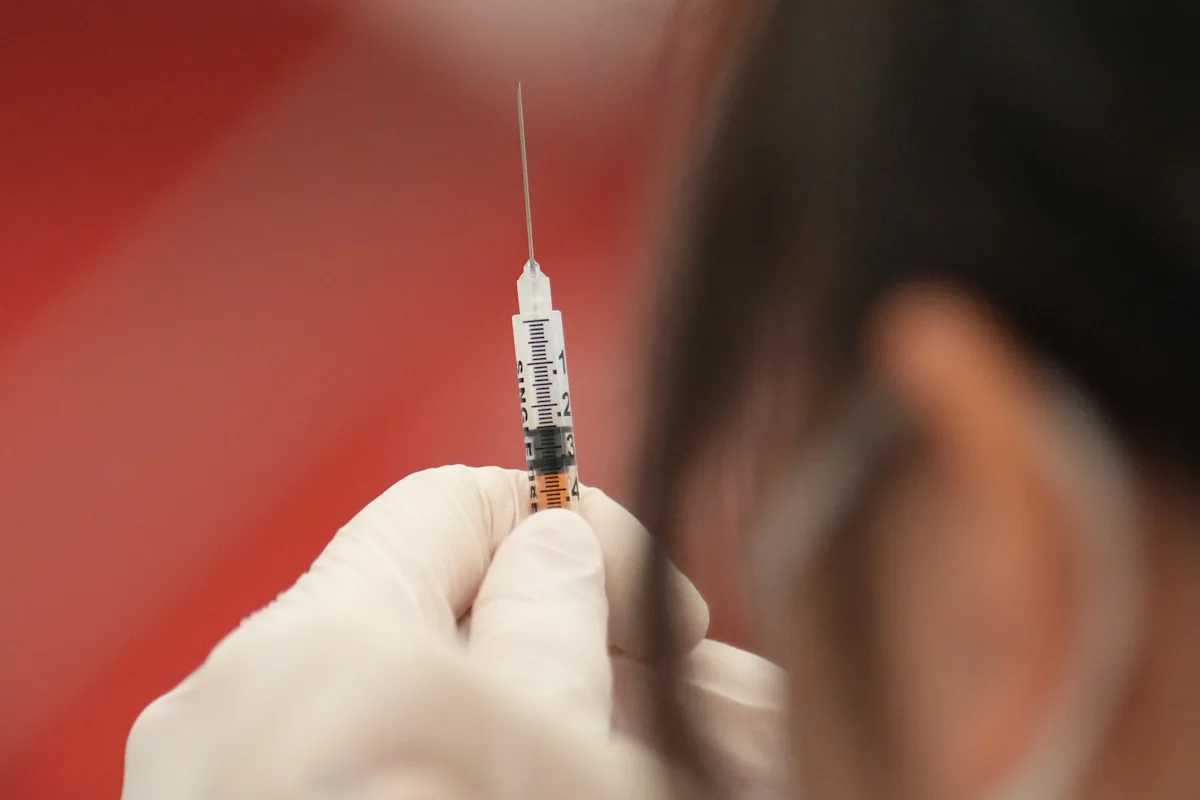
Shares of vaccine makers Moderna, Pfizer, and BioNTech fell after reports suggested US health officials might link Covid vaccines to the deaths of around two dozen children, based on VAERS data, amid upcoming CDC advisory meetings discussing vaccine safety and recommendations. The reports have heightened political tensions surrounding vaccine safety, despite scientific studies indicating rare serious side effects.