
Moderna's mRNA flu shot moves to FDA review after dispute resolution
Moderna says the FDA will review its experimental mRNA-based flu vaccine after resolving a public dispute, signaling movement toward potential regulatory consideration of the shot.
All articles tagged with #flu vaccine

Moderna says the FDA will review its experimental mRNA-based flu vaccine after resolving a public dispute, signaling movement toward potential regulatory consideration of the shot.

The FDA has agreed to review Moderna’s experimental mRNA flu vaccine after previously declining to file, with a decision due by August 5; Moderna seeks full approval for adults 50–64 and accelerated approval for 65+, contingent on a post‑marketing study, amid broader regulatory shifts and debate over vaccine oversight.

The FDA will review Moderna’s newly developed mRNA seasonal flu vaccine after a prior rejection over trial design, with no safety or efficacy concerns cited. Moderna says it has met with the FDA and proposed a revised regulatory pathway, seeking full approval for adults 50–64 and accelerated approval for 65+, potentially making the vaccine available for those 50+ in the 2026–2027 flu season if approved.

The FDA reversed its earlier decision and will review Moderna’s new mRNA flu vaccine for adults 50 and older, using a standard pathway for ages 50–64 and accelerated approval for those 65+, with a required post‑marketing study and a target review date of August 5.

The FDA declined to review Moderna’s experimental mRNA flu vaccine, citing an allegedly inadequate trial design for adults 65 and older that used a standard-dose control instead of a high-dose option, prompting critics to allege an anti-vaccine agenda and a chilling effect on vaccine development; Moderna and its supporters argue the design was approved and that higher-dose controls should have informed the analysis.

Childhood flu vaccine uptake fell 1.5 percentage points nationally as of Jan. 3, continuing a multi-year decline, amid a Kennedy-era overhaul of federal vaccine guidance that urges parents to consult doctors before vaccinating, drawing criticism from medical groups.
A preschool-age child in Virginia died from flu-related complications, the state’s first pediatric flu death this season. The Virginia Department of Health notes ongoing flu activity and low vaccination rates, urges eligible residents to get vaccinated, and highlights the presence of a highly contagious H3N2 subclade (subclade K) while vaccines remain protective against severe illness.
The US has revised its childhood vaccine schedule, now recommending fewer vaccines for flu, hepatitis A and B, and RSV, aligning with peer countries, and bypassing the usual approval process, under Health Secretary Robert F. Kennedy Jr.'s initiatives.

The CDC reports that flu activity in the U.S. has reached its highest level on record, with a significant rise in hospitalizations and cases, especially among children and young adults, driven by a new H3N2 subclade K strain. Despite the severity, vaccination rates remain low, and health officials warn that the peak of the flu season has not yet been reached, with ongoing circulation of COVID-19 and RSV.
Colorado is experiencing a severe flu season with rising hospitalizations, especially among children, partly due to a less effective vaccine match and a new aggressive strain. Despite some underreporting, data shows a significant increase in flu activity, and the flu currently poses a greater risk than COVID, emphasizing the importance of vaccination and preventive measures.
Flu hospitalizations in Colorado have surged to the highest level in over 20 years, driven by a new variant called subclade K and lower vaccination rates, with cases expected to rise further in the coming weeks.

Flu cases in North Carolina are surging earlier and more sharply this season, driven by the efficient spread of the subclade K variant, leading to increased hospitalizations and emphasizing the importance of vaccination and preventive measures.
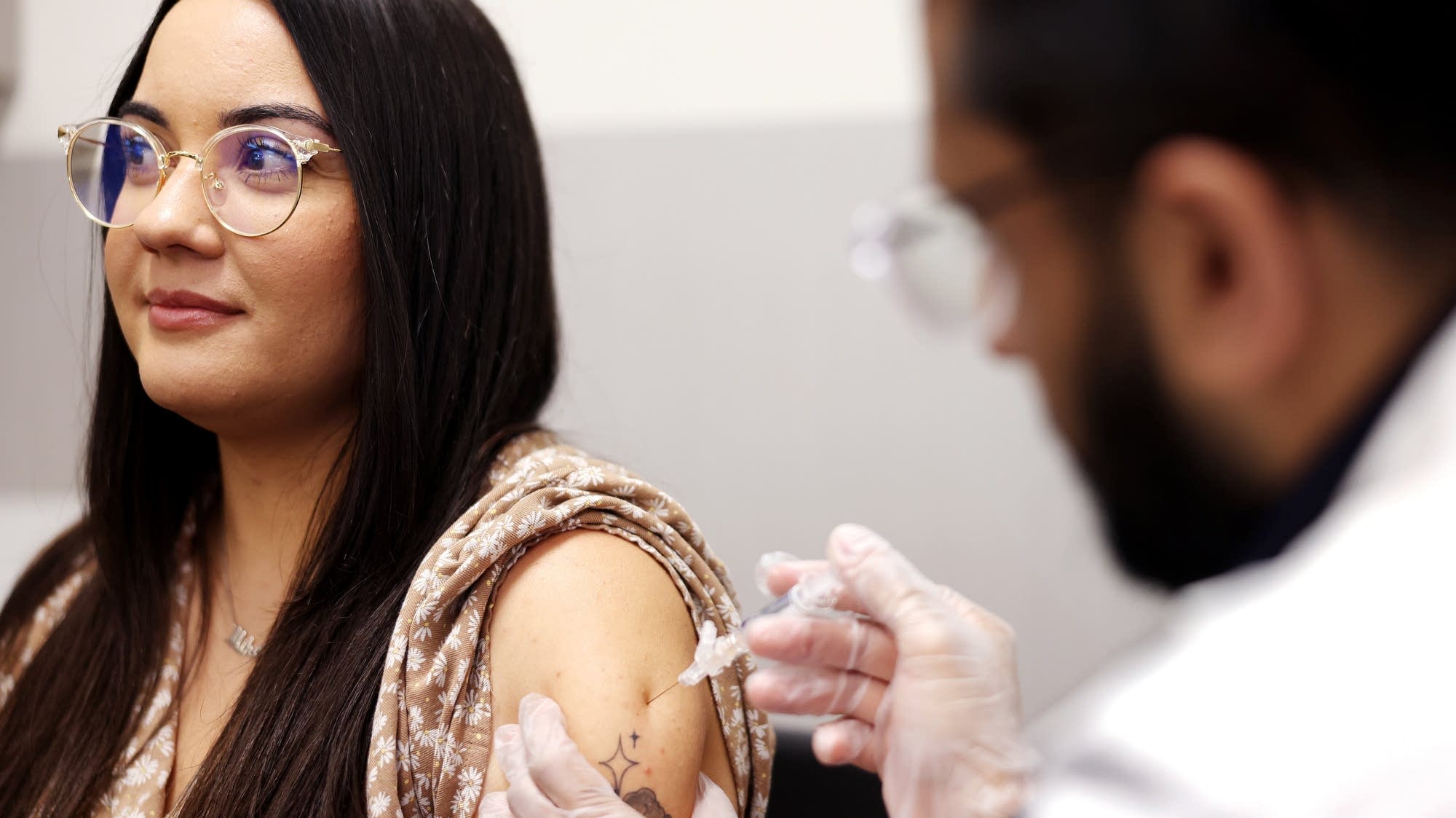
Flu cases are surging in Minnesota, with nearly 2,000 cases reported in December and more expected during the holidays, prompting health officials to recommend vaccination, masks, and avoiding social gatherings if symptomatic, especially for vulnerable populations like children and seniors.

A new 'super flu' strain, not covered by this year's flu vaccine, is spreading across the US, especially in New York, Louisiana, and Colorado, leading to increased hospitalizations. Symptoms include fever, chills, cough, sore throat, and muscle aches, with some cases resulting in severe complications like pneumonia. Experts recommend getting the flu shot despite its limited coverage, as it can help reduce symptoms.

The article discusses how reduced global influenza sample sharing, partly due to the US withdrawal from WHO, is impairing flu virus tracking and vaccine development, potentially leading to less effective flu shots and decreased pandemic preparedness.