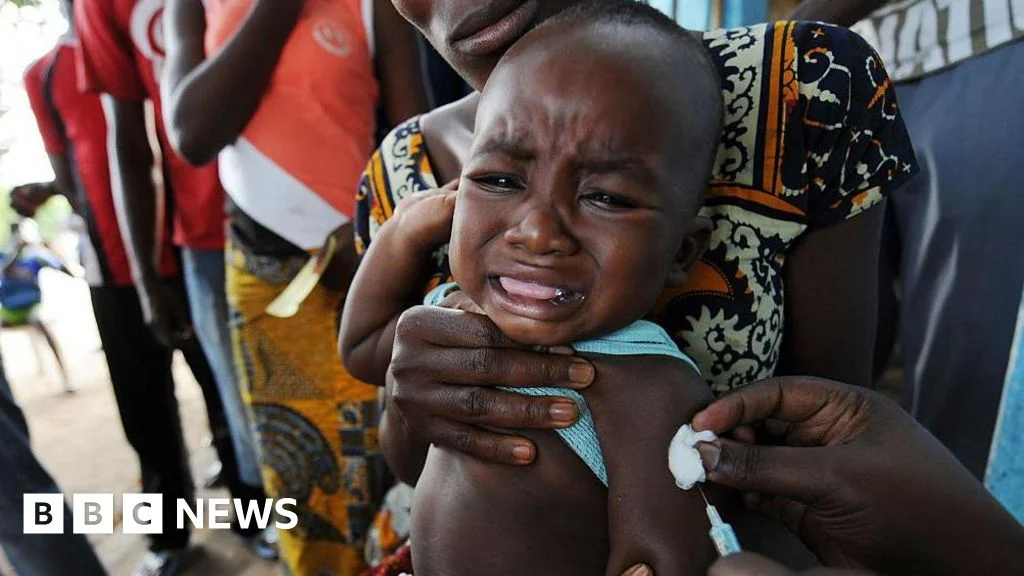
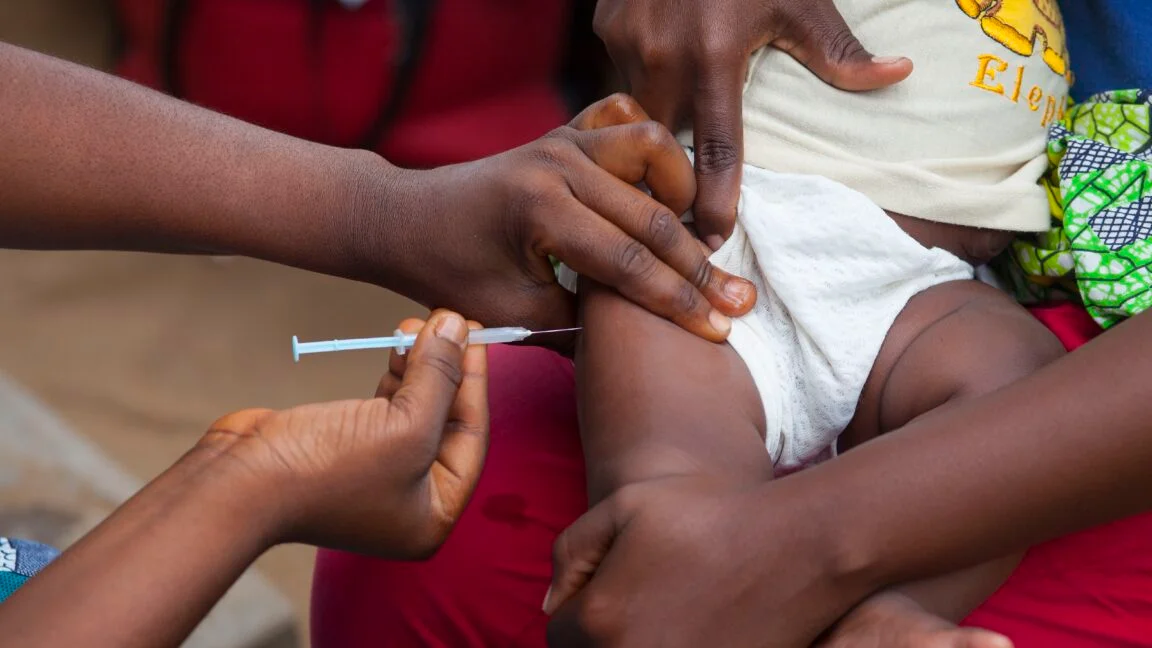
New Documents Trace Controversial Hepatitis B Birth-Dose Study's Shortcut to CDC Approval
A Rolling Stone investigation shows that a $1.6 million birth-dose hepatitis B vaccine study in Guinea-Bissau was moved directly from the CDC director’s office to grants management, bypassing normal scientific review and ethical oversight, amid pressure from Kennedy allies and irregular ethics approvals, drawing WHO concern and congressional scrutiny as the project remains in limbo.