WHO Denounces Birth-Dose Hepatitis B Trial in Guinea-Bissau as Unethical
TL;DR Summary
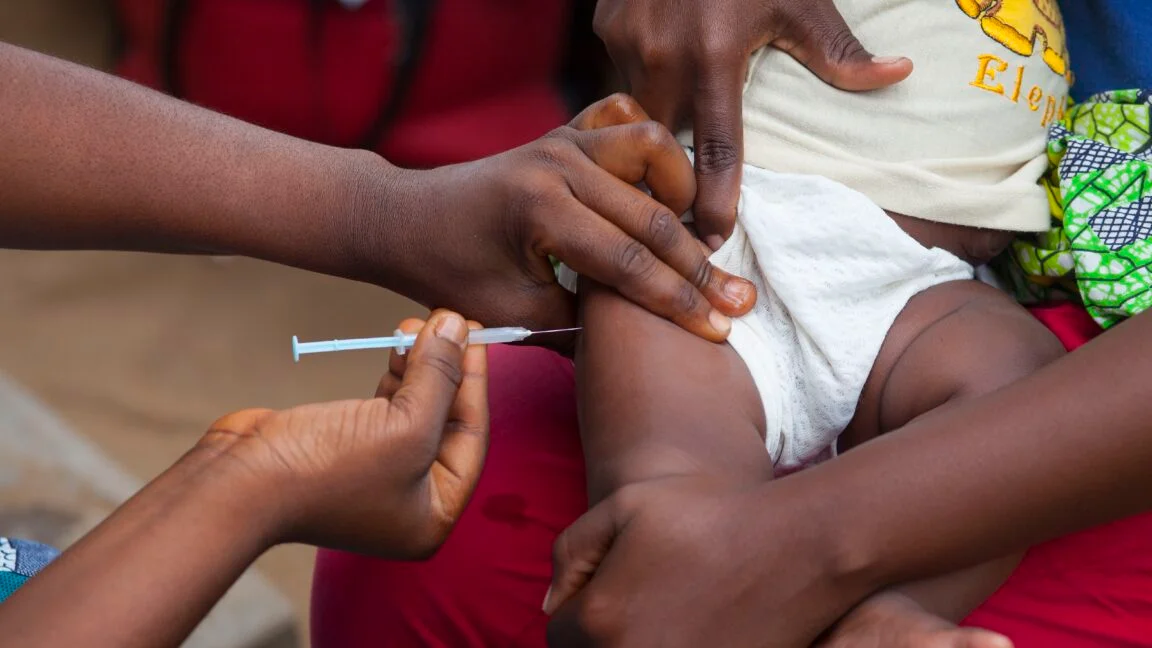
The World Health Organization condemned a US-funded trial in Guinea-Bissau that would randomize about 14,000 newborns to receive a hepatitis B vaccine at birth or at six weeks, calling the study unethical for withholding a proven, life-saving intervention. The trial, funded with $1.6 million by the CDC to Danish researchers Benn and Aaby, is suspended as Guinea-Bissau plans to adopt a birth-dose schedule in 2028 due to resource constraints, with concerns raised about potential bias and insufficient harm-reduction measures.
Topics:health#ethics-in-clinical-trials#guinea-bissau#hepatitis-b-vaccine#newborn-vaccination#world#world-health-organization
- WHO slams US-funded newborn vaccine trial as "unethical" Ars Technica
- Planned US-funded baby vaccine trial in Guinea-Bissau blasted by WHO BBC
- Statement on the planned hepatitis B birth dose vaccine trial in Guinea-Bissau World Health Organization (WHO)
- Guest post: The CDC hepatitis B study is unethical and must never be published Retraction Watch
- WHO director-general calls plans for U.S.-funded vaccine trial ‘unethical’ statnews.com
Reading Insights
Total Reads
0
Unique Readers
9
Time Saved
4 min
vs 5 min read
Condensed
92%
971 → 79 words
Want the full story? Read the original article
Read on Ars Technica