Guinea-Bissau pauses hepatitis B vaccine trial for ethics review
TL;DR Summary
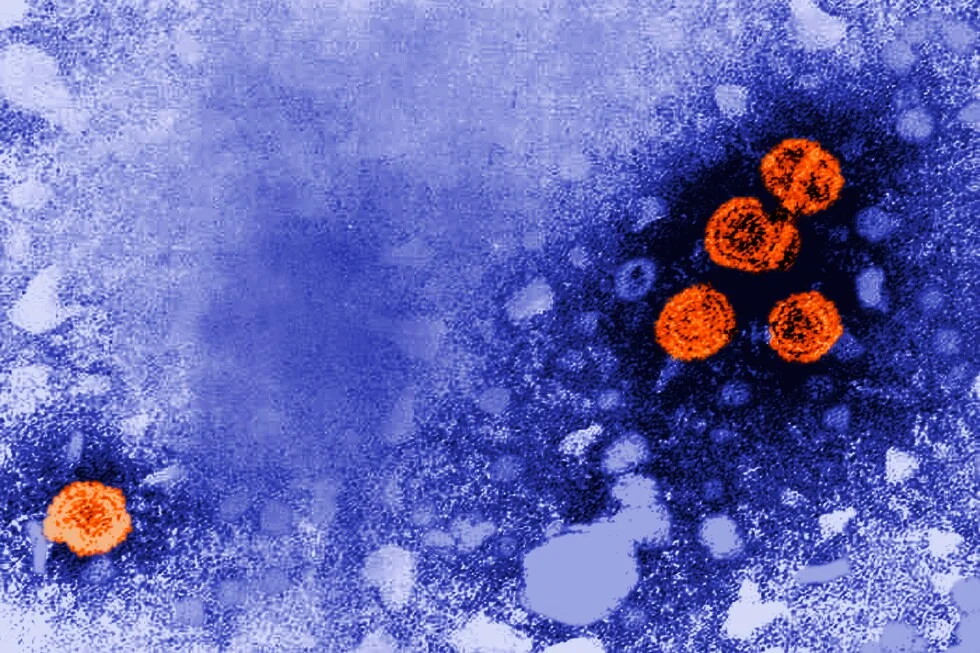
Guinea-Bissau has paused a hepatitis B vaccine study linked to supporters of former President Donald Trump to undergo an ethics review, delaying the trial as regulators assess safety and informed consent.
- Guinea-Bissau suspends Trump-backed hepatitis B vaccine study for ethical review AP News
- A vaccine trial is called 'unethical' and a 'unique' opportunity. Is it on or off? NPR
- Kennedy Plan to Test a Vaccine in West African Babies Is Blocked The New York Times
- Opinion | Trump’s HHS is backing a study in Africa that is drawing comparisons to the Tuskegee experiment MS NOW
- Controversial US study on hepatitis B vaccines in Africa ‘cancelled’ The Guardian
Reading Insights
Total Reads
0
Unique Readers
14
Time Saved
13 min
vs 14 min read
Condensed
99%
2,705 → 31 words
Want the full story? Read the original article
Read on AP News