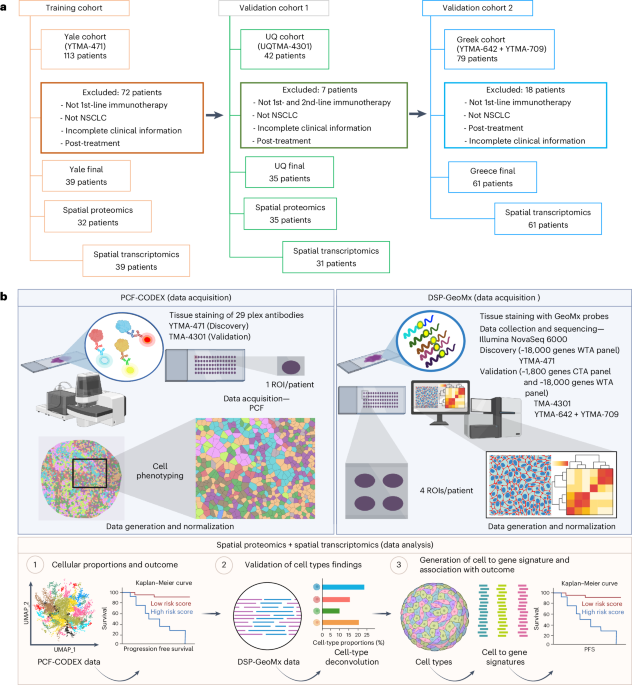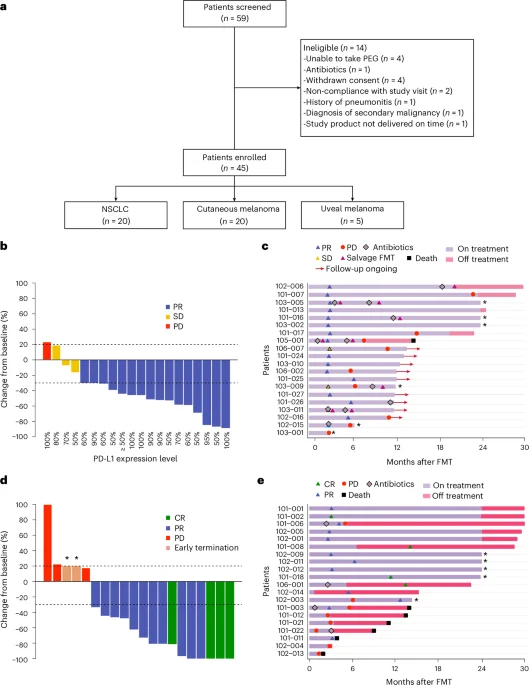
Healthy-donor fecal transplants boost frontline immunotherapy responses in NSCLC and melanoma
In the phase 2 FMT-LUMINate trial, healthy-donor fecal microbiota transplantation given before first-line anti-PD-1 therapy in NSCLC and anti-PD-1 plus anti-CTLA-4 in melanoma yielded high objective response rates (80% in NSCLC; 75% in melanoma) and was deemed safe overall, with no grade 3+ adverse events in NSCLC. Post-FMT microbiome shifts correlated with responses, including the loss of certain deleterious bacteria (e.g., Enterocloster and Clostridium species). Donor source did not predict efficacy, suggesting that remodeling the gut microbiome—specifically eliminating harmful taxa—drives the benefit. Preclinical mouse data supported this mechanism, showing that reintroducing the lost taxa abrogated the anti-tumor effects of immunotherapy. These findings support FMT as a strategy to overcome resistance to checkpoint inhibitors in NSCLC and melanoma.