Marburg virus glycoprotein engages NPC1 with a novel, high-affinity binding to boost entry
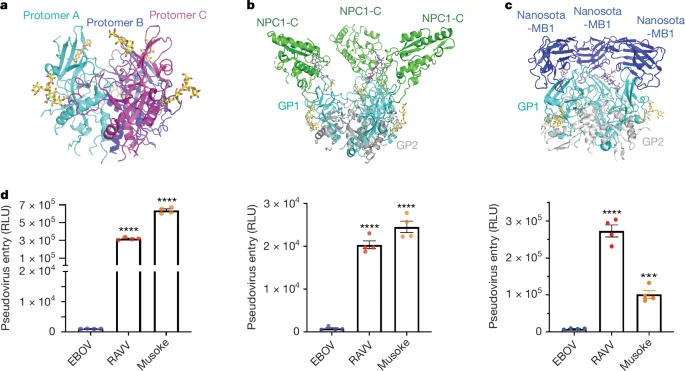
New cryo-EM structures of Marburg virus glycoprotein (MBV GP) in three states—unbound, NPC1-C bound, and nanobody-bound—reveal that MBV GP binds NPC1 with a distinct, higher-affinity orientation than Ebola GP, aided by a partially flexible glycan cap that blocks NPC1 only partially. NPC1 engagement induces substantial conformational changes in MBV GP that promote membrane fusion, explaining MBV GP’s markedly higher entry efficiency. A neutralizing nanobody, Nanosota-MB1, mimics NPC1 at the receptor-binding site and blocks NPC1 binding, neutralizing MBV pseudoviruses. Together, these findings illuminate MBV entry mechanisms and point to potential antiviral strategies targeting GP–NPC1 interactions and receptor-triggered transitions.
Reading Insights
1
8
49 min
vs 50 min read
99%
9,872 → 97 words
Want the full story? Read the original article
Read on Nature