FDA approves Rinvoq as first oral drug for Crohn's disease.
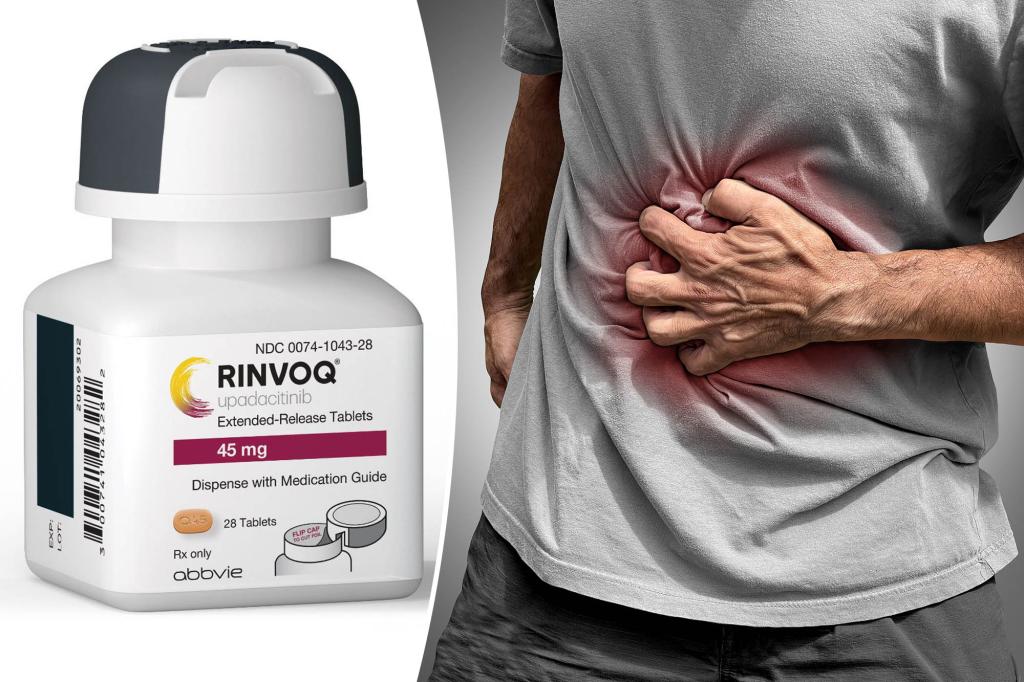
The FDA has approved Rinvoq, the first oral drug for moderate to severe Crohn’s disease, a chronic intestinal disorder. Rinvoq belongs to a class of drugs known as Janus kinase inhibitors, which moderate the body’s immune response and ease inflammation. The drug has already been approved to treat six other immune-related disorders including rheumatoid arthritis, the skin condition atopic dermatitis and ulcerative colitis. AbbVie Inc., the manufacturer of Rinvoq, is expected to rely on the drug as its leading moneymaker as the patents for its bestselling drug Humira are due to expire within a few years. However, the FDA has increased scrutiny of all Janus kinase inhibitors in recent years due to reports of serious heart-related side effects and cancer risks.
- What is Rinvoq? First oral drug for Crohn's disease approved by FDA New York Post
- With FDA nod for Crohn's, AbbVie wins 7th indication for Rinvoq FiercePharma
- FDA Approves Upadacitinib for Adults With Moderately to Severely Active Crohn Disease Pharmacy Times
- AbbVie gains Crohn's disease indication for Rinvoq in US (NYSE:ABBV) Seeking Alpha
- FDA Approves Upadacitinib (Rinvoq) for Crohn's Disease Medscape
Reading Insights
0
6
2 min
vs 3 min read
72%
439 → 121 words
Want the full story? Read the original article
Read on New York Post