"FDA Alert: Abiomed Heart Pump Recall Linked to 49 Deaths"
TL;DR Summary
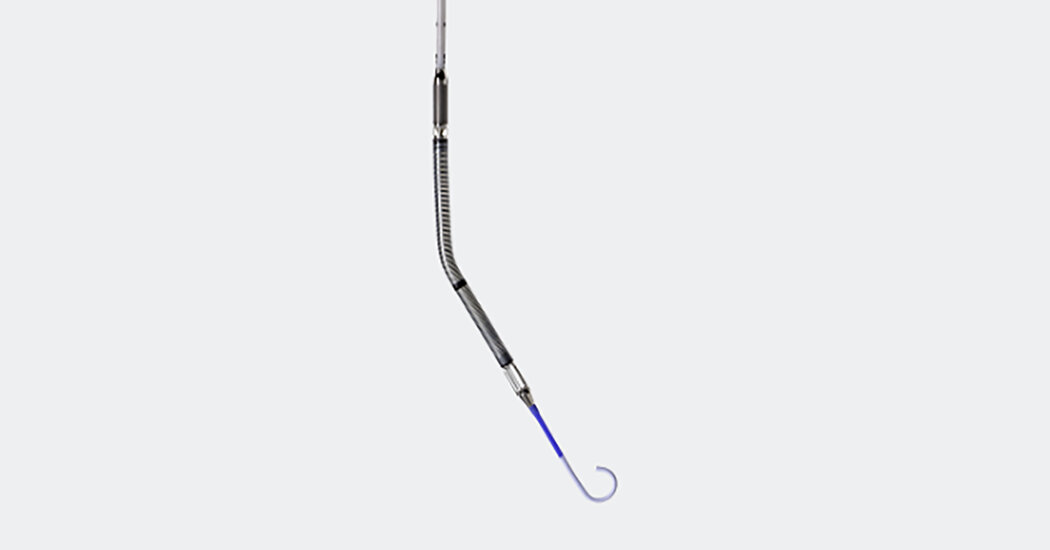
The FDA has issued an alert regarding the Impella heart pump, linking it to 49 deaths and numerous injuries, citing the risk of puncturing the heart's wall. Despite this, the device will remain in use. The manufacturer, Abiomed, was faulted for delayed notice of complications, and the FDA has received additional reports of heart-wall tears linked to patient deaths since the initial notice in 2021. This alert adds to concerns about the deadly side effects of cardiac devices, particularly those that take over the heart's blood-circulating function.
- FDA Issues Alert on Heart Pump Linked to Deaths The New York Times
- New warnings about heart pumps linked to 49 deaths CNN
- FDA shares new warnings over heart pumps that left 49 dead, over 100 seriously injured KOMO News
- J&J's Abiomed has a Class I Impella labeling recall with 49 deaths related to issue Mass Device
- Abiomed Impella Left-Sided Blood Pumps Recalled Medscape
Reading Insights
Total Reads
0
Unique Readers
5
Time Saved
2 min
vs 2 min read
Condensed
78%
390 → 87 words
Want the full story? Read the original article
Read on The New York Times